
Over the past 20 years, she has taught introductory and advanced courses in every division of the undergraduate chemistry curriculum, as well as interdisciplinary courses. Her Master’s and doctoral research involved the development of chemotherapeutic analogs of cisplatin and the development of instruments and methods for measuring ultra-trace concentrations of atmospheric sulfur compounds. in analytical chemistry at the University of Idaho. Julia Burdge did most of her undergraduate work at Iowa State University, completing her bachelor’s degree and Master’s degree in inorganic chemistry at the University of South Florida in Tampa. /PeriodicTable-56a12c983df78cf772682271.png)
Changes to this edition include specific refinements intended to augment the student-centered pedagogical features that continue to make this book effective and popular both with professors, and with their students.Ĭhapter 1 - Chemistry: The Science of ChangeĬhapter 3 - Quantum Theory and the Electronic Structure of AtomsĬhapter 4 - Periodic Trends of the ElementsĬhapter 7 - Molecular Geometry, Intermolecular Forces, and Bonding TheoriesĬhapter 9 - Chemical Reactions in Aqueous SolutionsĬhapter 10 - Energy Changes in Chemical ReactionsĬhapter 13 - Physical Properties of SolutionsĬhapter 18 - Acid-Base Equilibria and Solubility EquilibriaĬhapter 25 - ONLINE ONLY CHAPTER: Nonmetallic Elements and Their CompoundsĬhapter 26 - ONLINE ONLY CHAPTER: Metallurgy and the Chemistry of MetalsĪppendix 2 - Thermodynamic Data at 1 ATM and 25 degrees CĪppendix 3 - Solubility Product Constants at 25 degrees CĪppendix 4 - Dissociation Constants for Weak Acids and Bases at 25 degrees Cĭr. The fourth edition continues to build on the innovative success of the previous three editions. Far from a simple re-ordering of topics, this is a book that will truly meet the needs of the growing atoms-first market. Thus, the Atoms First approach empowers instructors to present the most complete and compelling story of general chemistry. Only after the study of matter and the atom will students have sufficient background to fully engage in topics such as stoichiometry, kinetics, equilibrium, and thermodynamics. 
Once mastery of the nature of atoms and electrons is achieved, the formation and properties of compounds are developed. This approach starts with the fundamental building block of matter, the atom, and uses it as the stepping-stone to understanding more complex chemistry topics. The Atoms First approach provides a consistent and logical method for teaching general chemistry. Can you fill in the names of all ten categories of elements just by going by the symbols of elements in those categories? Which elements belong to which categories? Use colored markers to outline the elements and their categories in the same color.Julia Burdge and Jason Overby Chemistry: Atoms First 4 Janu9781260240696 Hide element names and symbols and challenge students to complete the table. Just open the file in a PDF reader supporting layers and begin customizing! Show all layers for a complete view including groups, periods, legends, colored outlines, corners (for black and white printing), as well as the usual atomic number, weight, symbol, and name. Letters in element box corners show categories so no information is lost with black and white printing. 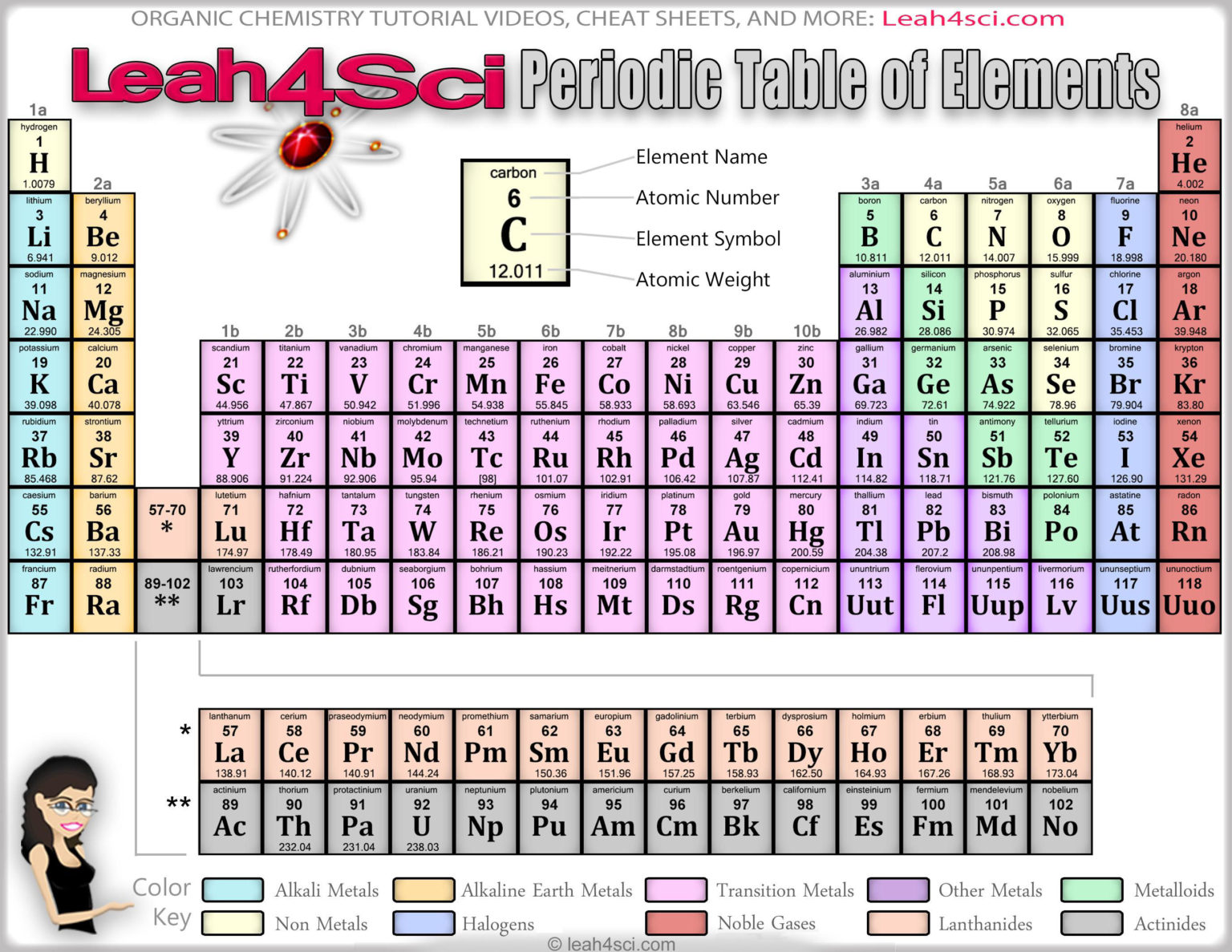
Colored outlines rather than filled blocks save ink.Fill-in worksheets for students to build their own periodic table.Handouts can provide only necessary information during test taking.Create quizzes by hiding names or categories.Hide unnecessary or grade-level inappropriate information.Ptable's new, up-to-date periodic table PDF and wide periodic table PDF are layered so you can choose exactly what you want to print, and are the perfect companion to the periodic table classroom poster. Printable Periodic Table PDF - Ptable Printable Periodic Table PDF
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
